|
|
 |
The tethered cord syndrome is a complex
developmental malformation, with the underlying pathological
anomaly being a dura mater defect or dural schisis. The dural
schisis may not be the only developmental defect, but it is
probably the most basic one and one that occurs more commonly
than is generally recognized. Establishing the diagnosis and
assessing the extent of functional disability is often difficult
but may be aided considerably by the use of several clinical neurophysiological
studies, including somatosensory evoked potentials (SSEPs), urodynamics with sphincter and
pelvic floor electromyography (EMG) and anal sphincter EMC and
pressure monitoring for intraoperative use.
Developmental Anatomy
The spinal cord is freer within the vertebral
canal than is the brain within the cranium. The spinal dura
mater is composed of dense connective tissue with few elastic
elements derived from paraxial mesoderm. It is separated from
the vertebral internal periosteum by the epidural space, which
contains fat cells, blood vessels, and loose connective tissue.
The spinal cord needs to be completely free from the vertebral
column during development because the rates of growth of the two
structures are different. Early in development, the caudal
region of the spinal cord undergoes a progressive upward
displacement or retrogression relative to the caudal vertebral
column. The conus medullaris, which is initially at the
coccygeal level in the 30-mm embryo, ascends through the S4
level in the 67-mm embryo, to the L3 level by birth (40 weeks conceptional age), and to the adult L1-2 level by 49 to 50
weeks conceptional age.
The subarachnoid space elongates progressively to accommodate
the elongating spinal nerve roots and filum terminale. The filum
terminale must also elongate because the cord retains its
original coccygeal attachment through this structure. Early
dural schisis (below L3) through which the spinal cord comes in
direct contact with subcutaneous tissue lends to tethering of the spinal cord to this tissue. Later, subcutaneous
adipose tissue penetrates and expands into the intraspinal
space. This results in a low conus medullaris and a short,
thick filum terminale. It is possible that the adipose tissue is
stimulated by its direct contact with neural elements and the
abundant arachnoidal vascularity through the dural schisis.
Neuroanatomy of Somatosensory Evoked Potentials
Evoked potentials recorded from the body's surface are either
near field or far field in nature, that is, the generator source
is close or distant to the site of recording. The generators may
be in gray matter or white matter. Generators in gray matter
produce postsynaptic potentials (PSPs), which may be near field
or far field. Near field PSPs are probably responsible for
cortical components of SSEPs.
White matter generates compound action potentials (APs), which
are propagated through fiber tracts. The latencies of the
propagated APs increase proportionately to the distance from the
point of stimulation and hence are dependent on the recording
electrode position. These are recorded only in close proximation
to the fiber tract itself and thus are termed "near field
potentials" (NFPs). Because they are close to the site of
origin, the amplitude is relatively large (> 1
µV). Other
evoked potentials may be recorded at long distances from the
point of propagation and are generated when a travelling impulse
(signal) passes through a certain anatomical site or fixed point
along the nerve. These are called "far field potentials" (FFPs).
It was previously considered that FFPs reflected the approaching
volley recorded beyond the point of termination of an active
fiber. More recently, it has been suggested that FFPs are
generated because of abrupt changes in the geometry of tissue
surrounding the nerve, a change in the medium through which the
volley is transmitted, or a change in the direction of the
fibers.
In summary, NFPs have a specific distribution (topographic
specificity), latencies that
vary according to the recording electrode placement, amplitudes>
1 µV, and generally negative polarity. FFPs have a diffuse
distribution, fixed latencies, amplitudes <1
µV, and polarity
that probably reflects a volume-conducted positivity.
Potentials are labelled according to polarity and mean latency
from a sample of the normal population. As one would expect,
latencies change with body growth and nervous system maturation.
Hence, labels differ between children and adults.
Posterior Tibial Nerve Somatosensory Evoked Potentials
There are less standard evoked potential component designations
for posterior tibial nerve (PTN) SSEPs than for median nerve
SSEPS. For the purpose of discussion of generators of SSEP-PTNs,
adult terminology is used, with child or infant notation
following in parentheses. Following PTN stimulation, electrodes
over the popliteal fossa record the electronegative peripheral
nerve action potential N8 (N5). Electrodes over the lower spine
record two electronegative potentials: the N19 (N11) and the
N22 (N14). The N19 (N11) represents the afferent volley in the
cauda equina. The N22 (N14) is a stationary potential and
probably reflects postsynaptic activity of internuncial neurons
in the gray matter of the spinal cord. Electrodes over the
cervical spine record another later stationary potential: the
N29 (N20). This component may reflect postsynaptic activity in
the nucleus gracilis. The P37 (P28) is the first major
localized recorded component on the scalp. It reflects the
ipsilaterally oriented cortical surface electropositivity, while
the electronegative end of the dipole may be recorded
contralaterally. There is a great deal of intersubject
variability in the topography of the P37 (P28) in adults and
especially in children. This is probably related to the known
anatomical difference in the location of the primary sensory
area for the leg. When the leg area is located at the superior
edge of the interhemispheric fissure, the cortical generator for
P37 (P28) is vertically oriented and its amplitude is maximally
close to the vertex. When the leg area is located more deeply in
the fissure, the cortical generator is more horizontally turned
and the P37 (P28) projects ipsilaterally.
Pathophysiology of Tethered Cord Syndrome
Four decades ago, the usual explanation for the neurological
deficit associated with tethered cord syndrome was the effect of
traction in preventing the ascent of the spinal cord within the
spinal canal during growth. However, Barson pointed out in 1970
that the spinal cord does not ascend significantly after birth.
The incongruity in observations is due to the fact that the
spine grows most rapidly during embryogenesis and after puberty
(during teenage growth spurts), while symptoms of tethering most
often are observed in early childhood (age 3-10 years). James
and Lassman reported a clinical case in which, during a postmortem examination, a small bony septum from the midline of
the laminae of L3-4 to the underlying vertebral
body was found in an aged woman. She had never had any
neurological deficits. Had there been significant ascension of
the spinal cord after birth, she would have had to have had
neurological deficits, so the authors argued. While this was
not necessarily so because issues of the divided cord segments
rejoining below the spur and the size of the cleft between cord
segments (small or large) were not addressed, the concept that
the spinal cord ascended postnatally and produced neurological
deficits by traction alone in tethered cord syndrome seemed
untenable.
|
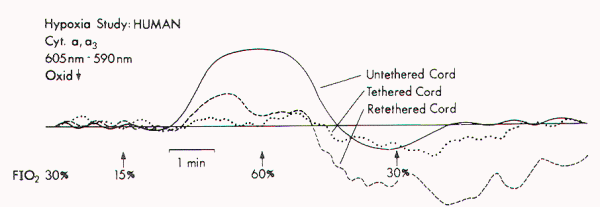
Yamada et al examined the mitochondrial oxidative metabolic
changes in the spinal cord before and after subjecting it to
stretching. Using reflection spectrophotometry, they monitored
in vivo changes in the reduction: oxidation (redox) ratio of
cytochrome a, a3 in animal models and in human tethered spinal
cords (Figure 1). They found a marked metabolic and
electrophysiological susceptibility of the lumbosacral cord
subjected to hypoxic conditions, especially under traction with
hypoxic stress
(Figure 2). They concluded that symptoms and signs of tethered
spinal cord were associated with lumbosacral neuronal
dysfunction and that this dysfunction is possibly due to
impairment of mitochondrial oxidative metabolism. This is
supported by the associated evoked potential changes (see
below). Most paediatric neurosurgeons now believe that the
chronic stretch on the cord produced by tethering is an
essential part of the problem but that superimposed insults such
as acute flexion episodes or cord hypoxia are also needed for
symptoms to become manifest. |
 |
 |
| Figure 1. Redox changes during hypoxia in one group of the human
tethered cords (Type 1). No redox change is seen before
untethering (dotted line), but a reduction similar to that in
normal cat cords is noted after untethering (solid line). No
reduction occurs while the cord is temporarily retethered
(interrupted line). FIO2 = fraction of inspired oxygen.
(Reproduced from Yamada et al) |
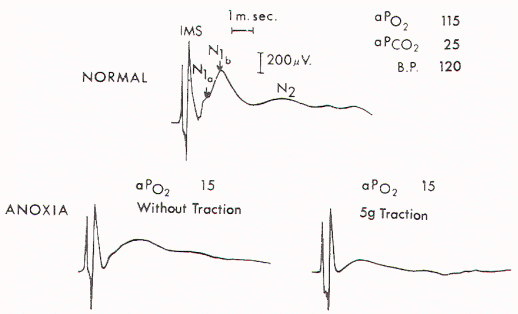
Figure 2. Upper) Normal cord potentials in response to dorsa!
root stimulations. IMS: from the posterior column; N1a: from
the afferent terminals; N1b: from the interneurons of the first
order; N2: from the interneurons of the second and third orders.
Lower) Marked change in the cord with traction of 5 gm.
(Reproduced from Yamada et al) |
|
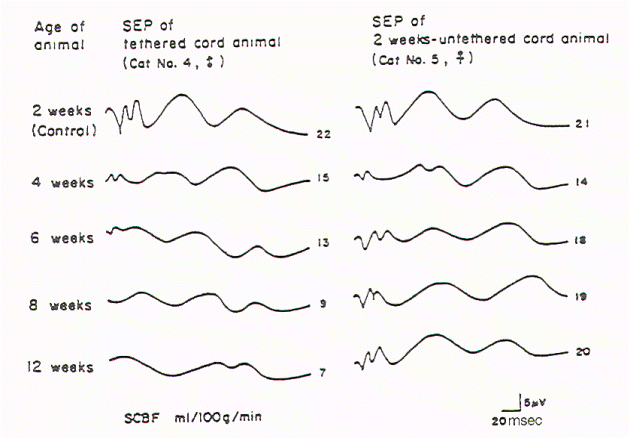
Kang et al tethered and untethered the cords of immature
kittens and studied the effects of these manipulations on
regional spinal cord blood flow, clinical features, and SSEPs.
They found that cord tethering caused a reduction of regional
spinal cord blood flow in the distal spinal cord close to the
site of tethering. The reduction in regional spinal cord blood
flow (rSCBF) became progressively worse over the weeks following
the tethering (Figure 3). Untethering of the cord led to an
increase in the rSCBF if the untethering occurred by 2 weeks
after tethering. Delaying the tethering 8 weeks
prevented the return to the normal level of the rSCBF. Changes
in the evoked potential occurred when rSCBF fell below 14 ml/100
g/ min. The decrease in rSCBF had occurred by 2 weeks after
tethering. |
 |
| |
Fig-3 |
|
Diagnostic Clinical Neurophysiological Studies
Electrophysiological studies that help with diagnostic
formulation include SSEPs after peroneal nerve stimulation
(SSEP-PN), after pudendal nerve stimulation (SSEP-PuN) and after
posterior tibial nerve stimulation (SSEP-PTN), bulbocavernosus
reflex responses (BCR), and urodynamics with sphincter and
pelvic floor EMG.
Somatosensory Evoked Potentials
It has been three decades since SSEPs were first used to evaluate
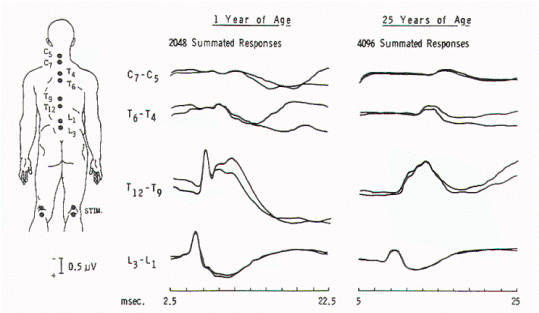
patients with occult spinal dysraphism. Cracco and Cracco
recorded scalp and spinal responses after peroneal stimulation
over the cauda equina and rostral spinal cord in adult and child
control subjects (Figure 4).
These spinal potentials consisted of low-amplitude triphasic
waves over the cauda equina and larger potentials over the
caudal spinal cord. Scalp potentials had latencies of 30 to 34
msec for electropositive components and 40 to 45 msec for
electronegative components. In patients with sacral lipomas and
no or minimal neurological findings, spinal potentials normally
recorded over the lower thoracic spine (T9-12) were recorded
over the lumbar spine, suggesting caudal displacement of the
spinal cord (Figure 5). In children with more extensive
neurological findings (foot deformities, neurogenic bladders),
relatively normal potentials were recorded over the cauda
equina, and cerebral potentials were absent. Others have
subsequently verified the diagnostic value of SSEPs.
|
 |
 |
| Fig-4 |
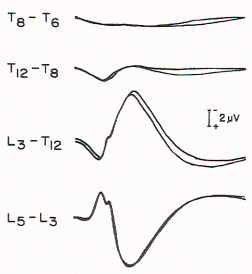
Figure 5. Spinal responses in a 3-year-old child with
thoracolumbar myelomeningocele. The large complex response that
is recorded over T12 to T9 in normal children is present over L3
in this child, suggesting caudal displacement of the spinal
cord. |
One group reported a patient who had postoperative SSEP-PN
studies that were slightly improved compared to preoperative
studies, and the patient had improved clinically.
The authors have systematically studied children and young
adults with tethered cord syndrome using SSEP-PTNs. Because
SSEP-PN scalp and spine components are lower in amplitude than
those produced by PTN stimulation and because the topography of
the scalp component of SSEP-PN is more variable than that of
SSEP-PTN, SSEP-PTNs were used rather than SSEP-PNs for
evaluation in children suspected of having tethered cord
syndrome. Clinical, myelographic, and operative studies were
prospectively evaluated in 22 consecutive patients, aged 18
months to 22 years, with symptoms of tethered cord syndrome.
Ten had previously undergone repair of lumbosacral meningomyelocele. In 19 patients, the diagnosis was established
radiologically and/ or intraoperatively. In three patients with
clinical symptoms but without radiographically demonstrable
lesions, SSEP-PTNs were normal.
| Details of SSEP-PTN methodology have been reported elsewhere.
Briefly, square wave stimuli are delivered to the PTN at the
ankle, with an intensity sufficient to cause a twitch of the
abductor hallucis muscle or three times the sensory threshold.
If the patient were anesthetic to the stimulus, then a sensory
threshold three times that of a similarly aged control subject
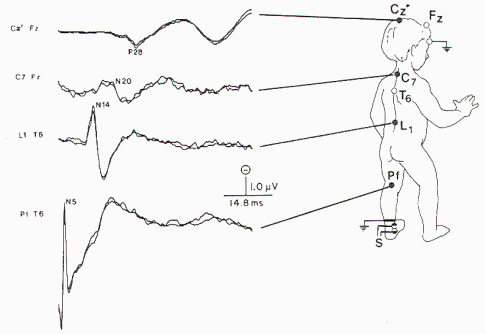
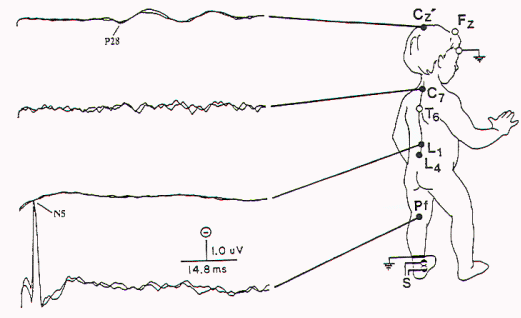
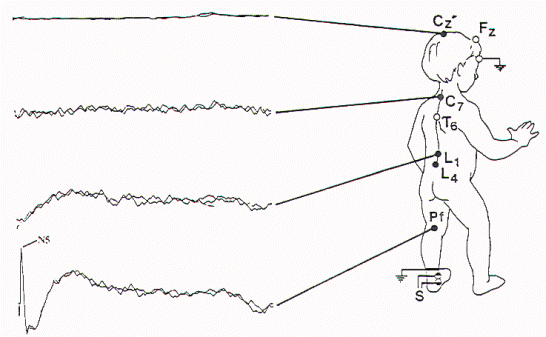
was used. The recording montage is presented in Figure 6. The bandpass
was 30 to 1500 Hz, with 40,000 amplification.
Between 1000 and 2000 responses were averaged and
replicated. Normative data are largely based on
height. Only occasionally will age be used because
generally the authors believe height is a better
predictor of peak latency (Figure 7). |
 |
 |
| |
|
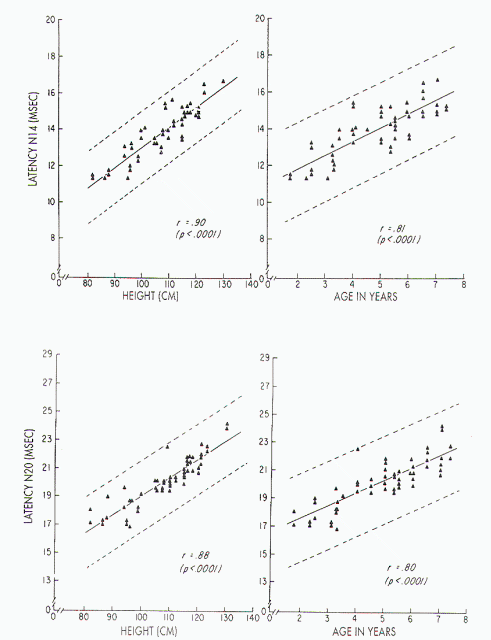
Figure 7. Upper left) Relationship between stature and absolute
latency of N14 in children, with height ranging from 82 to 130
cm: x = 1.84 ± 0.11 (height). Upper right) Relationship between
age and absolute latency of N14 in children aged 1-8 years: x =
10.26 ± 0.74 (age). Lower left) Relationship between stature and
absolute latency of N20 in children, with height ranging from 82
to 130 cm: x = 4.60 ± 0.14 (height). Lower right) Relationship
between age and absolute latency of N20 in children aged 1-8
years: x= 15-51 ± 0.95 (age). (Reproduced from Gilmore et al16
with permission.) |
Based upon the absence or presence and the latency of N22 (N14)
and P37 (P28), a severity rating scale for SSEP-PTN was
developed (Table 1). The generator of the N22 (N14) is the
lumbar spinal cord gray matter (Table 2). Thus, the lumbosacral
neuronal dysfunction reported by Yamada et al might be reflected
especially in abnormalities of the N22 (N14).
|
Table 1. Severity
Rating Scale for SSEP-PTN |
| Severity
Score |
N22
(N14) Latency |
N22
(N14) Amplitude |
P37 (P28)
Latency |
| Severely abnormal |
| 1 |
absent |
absent |
absent |
| 2 |
normal |
decreased |
absent |
| 3 |
delayed |
normal |
absent |
| 4 |
absent |
absent |
delayed |
| Moderately abnormal |
| 5 |
absent |
absent |
normal |
| 5 |
delayed |
normal |
normal |
| 6 |
normal |
decreased |
delayed |
| 7 |
normal |
normal |
delayed |
| Mildly abnormal |
| 8 |
normal |
decreased |
normal |
| 9 |
normal |
decreased |
normal |
| 10 |
normal |
normal |
normal |
|
Table 2. Presumed Generators of
SSEP-PTN |
| Component |
Origin |
| N8(N5) |
tibial
nerve action potential |
| N19(N11) |
cauda equina |
| N22 (N14) |
lumbar cord
gray matter |
| N29 (N20) |
nucleus gracilis |
| P37 (P28) |
mesial sensory cortex |
|
Table 3. Severity Scale for Clinical Assessment of Tethered Cord
Syndrome |
| Gait |
Bowel/bladder history |
| 0 - unable
to walk unassisted |
0 - total incontinence |
| 1 - severe bilateral deficit |
1 -
intermittent incontinence, uncontrolled |
| 2 - severe unilateral deficit |
2 - intermittent
incontinence, controlled |
| 3 - mild
bi- or unilateral deficit |
3 - increased frequency |
| 4 - walks normally |
4 - total control |
|
Sensory (pinprick) |
|
0 - no sensation |
|
1 - diminished sensation |
| 2 -full sensation |
|
Lower limb strength |
|
Use clinical scale of 0/5-5/5 for weakest joint on
the limb |
|
Deep
tendon reflexes |
| The sum of the ankle and knee score for each lower limb
(possible 4+ at each joint) |
Yamada developed clinical severity scales using the factors
of gait, bowel/bladder continence, motor, sensation, and deep
tendon reflexes (Table 3), and also an operative severity scale
based on the presence of lipoma, tension on the filum terminale,
and/or extent of adhesions and cord movement after lysis of
adhesions (Table 4). In three patients with clinical symptoms
but without radiographically demonstrable lesions, SSEP-PTNs
were normal. In the 19 patients with tethered cord syndrome, the
clinical score and SSEP-PTN score correlated significantly
(r=.81, P<0.001). The location and direction of the tethering
structures influenced the SSEP-PTN findings (Table 5). In patients
with involvement primarily of the conus, the N22 (N14) was
present but diminished in amplitude. In patients with extensive
attachment of the spinal cord, the N22 (N14) was generally
absent (Figure 8) and frequently the N19 (N11) was also
absent. Patients with scalp SSEP-PTN asymmetry tended to have
the more severe abnormality contralateral to cord deviation or
rotation (Figure 9).
|
Table 4. Severity Scale for
Operative Findings in Tethered Cord Patients |
|
Lipoma |
| |
0- no lipoma |
| |
1 - lipoma
not extensively attached |
| |
2 - lipoma extensively
attached |
|
Filum terminale |
| |
0- flaccid
|
| |
1 -
moderately tight |
| |
2 - very tight |
|
Adhesion extent |
|
0 - only filum terminale attachment |
| |
1 - loose
attachment in addition to filum |
| |
2 - extensive, tight
adhesions |
|
Cord movement |
| |
Upward movement of the cord after lysis of adhesions (cm) |
|
Table 5. Clinical and Operative Severity Scores for Patients
with Different Degrees of SSEP-PTN Abnormalities |
| SSEP-PTN
Abnormality |
No. of
Patients |
No. of Studies |
Clinical
Score |
Operative Score |
| Mild |
12 |
20 |
15.9±1.4 |
1.3±0.6 |
| Moderate |
9 |
12 |
9.9±2.0 |
3.1±1.1
|
| Severe
|
5 |
7
|
8.9±1.3 |
4.1±1.0
|
 |
 |
| Figure 8. Abnormal SSEP-PTN with absent N14 (lumbar potential)
and N20 (cervical potential) in a child with extensive
attachment of the spinal cord. |
Figure 9. Abnormal SSEP-PTN. This is more abnormal than the
study in Figure 8 in that the lumbar, cervical, and cortical
potentials are all absent. The more severe abnormality was
contralateral to the spinal cord deviation, with the P28
(cortical potential) absent after left leg stimulation. |
|
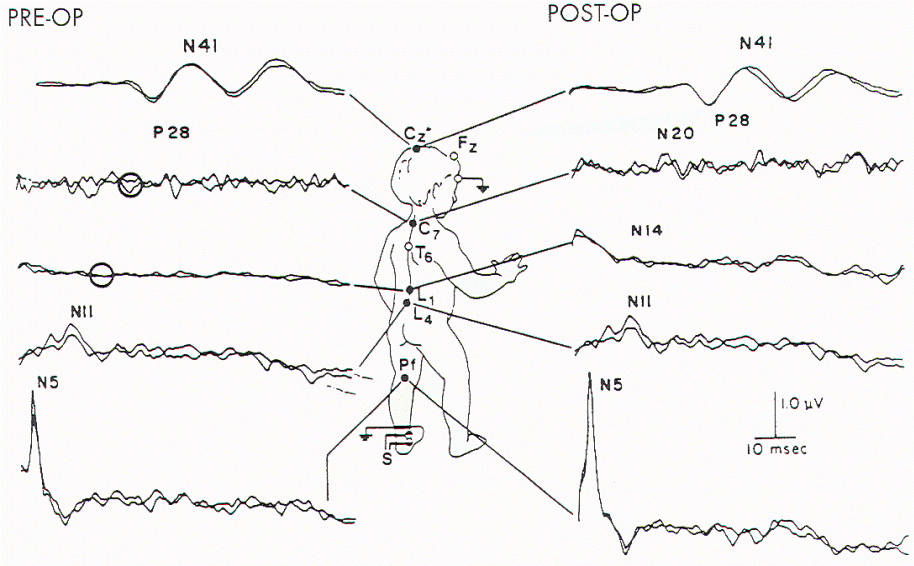 |
| Figure 10. Preoperative and
postoperative SSEP-PTN in a child who was found at
surgery to have extensive tethering and rotation of
the sacral spinal cord. Both N14 and N20 (lumbar and
cervical spinal cord evoked potentials,
respectively) were absent prior to operation
(circles) and appeared postoperatively. |
Postoperative SSEP-PTNs were sometimes
improved (Figure 10 and Table 6). Findings associated with
clinical improvement include an increase in the amplitude of N22
(N14), normalization of the P37 (P28):N22 (N14) amplitude ratio,
shortening of the N22 (N14) latency, appearance of previously
absent N22 (N14), and a decrease in central conduction time
(latency P37 (P28) -latency N22 (N14)). Some technical points
were important: children 8 years old should be tested in the
waking state because latency of the scalp component of SSEP-PTN
varies with that state.
The N14 in children (probably generated by structures generating
N22 in adults) is considerably higher in amplitude than in
adults. Hence, its absence is a more reliable indicator of
dysfunction in children.
|
Table 6. Relationship Between Postoperative Clinical Improvement
Score and Specific SSEP Changes |
| |
N22 (N14) Appearance |
N22(N14) Increased Amplitude |
N22 (N14) - P37 (P28) Latency Decreased |
| No. of
patients |
4 |
3 |
4 |
| Mean clinical improvement |
| score |
2.3 |
1.3 |
2 |
There is a small but growing literature on evaluating the spinal
cord with SSEP-PuN. None of these studies have
systematically evaluated the use of SSEP-PuN specifically in
patients with tethered cord syndrome, but application to this
clinical condition is obvious. The technique consists of
stimulating any of several structures innervated by the pudendal
nerve. The most accessible structure is the dorsal nerve of the
penis, which can be stimulated bilaterally using ring electrodes
placed at the base of the penis or unilaterally using laterally
placed cup electrodes. Stimulation of the urethra or anus is
possible using catheter electrodes with tip-inflatable balloons
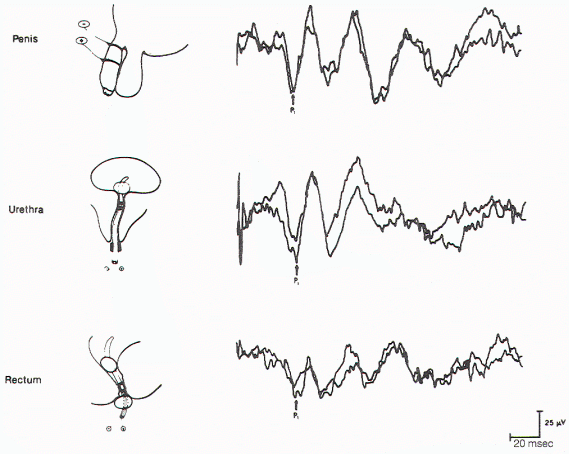
to maintain appropriate stimulus localization (Figure 11).
Square wave electrical stimuli of 0.3 msec applied at 1.7 to 3.1
Hz with an intensity 2.5 times threshold are delivered. Scalp
recording electrodes are placed at Cz (2 cm behind Cz,
International 10-20 Electrode Placement System) and Fpz. Spinal
electrodes are placed at the spinous process of T12 or L1 with
reference to electrodes at the iliac crest or T6 spinous
process. A common bandpass is 10 to 500 Hz. Sampling time ranges
from 100 to 200 msec. Five hundred to 1000 responses are needed
for the critical response and 1000 to 2000 responses for the
spinal response. Only stimulation of the dorsal nerve of the
penis will elicit a spinal response; stimulation of other
structures innervated by the pudendal nerve will not elicit a
spinal response. The mean latencies of the cortical response of
the SSEP-PuN is similar to, but slightly longer than, that of
the cortical response of the SSEP-PTN: 42.3 + 1.9 msec.
Amplitude varies depending upon which branch of the PuN is
stimulated, The spinal response has a latency of 12.9 + 0.8
msec. This latency is much slower than that of the spinal
response of the SSEP-PTN, resulting in a long central conduction
time (cortical peak latency spinal peak latency) of
approximately 30 msec. This has been attributed to central
conduction via smaller fibers than those giving rise to the
SSEP-PTN response or to a greater number of synaptic connections
in the pathway.
With spinal cord lesions at or above TI2, there may be absence
or prolongation of the cortical potential. With spinal cord
lesions at L1 or below, there will be absence or prolongation of
the lumbar potential as well as the cortical potential. In
actual practice, the value of this localization is limited
because obesity, peripheral neuropathy, or stimulation of the
PuN at sites other than the dorsal nerve of the penis will also
lead to absence of the spinal potential.
 |
 |
| Figure 11. Cortical evoked
responses on stimulation of the dorsal nerve of the
penis (upper), urethra (middle), and anal sphincter
muscle (lower) in the same individual. |
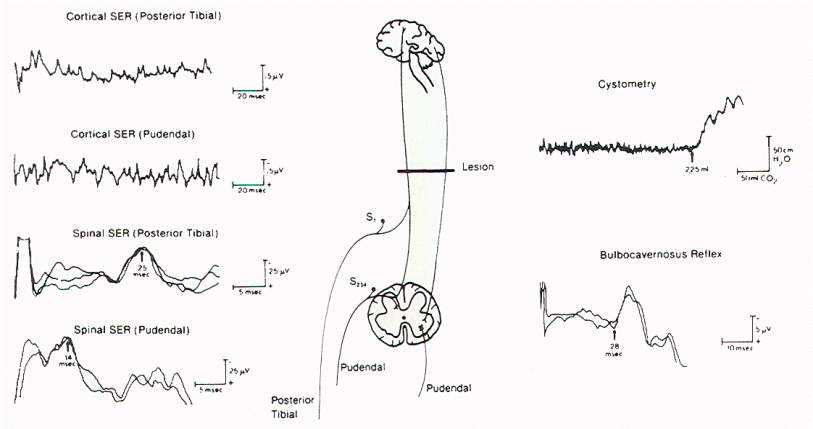
Figure 12. BCR responses
recorded from different sites in the perineum on
stimulation of the dorsal nerve of the penis. SER =
sensory evoked response. |
Bulbocavernosus Reflex
The BCR is elicited by squeezing the glans penis or glans
clitoridis or pulling on an indwelling Foley catheter. The
response, contraction of the external anal sphincter muscle, may
be seen or palpated. The absence of a response in a man is
highly suggestive of a neurological lesion. Unfortunately, it is
absent in up to 20% of normal females.
The corresponding BCR electrophysiological potential is recorded
over the perineum using small surface electrodes placed midway
between the penis (or vagina) and anus. Stimulation electrode
placements and parameters are similar to those of SSEP-PuN.
Recording parameters are also similar, except that 30 to 100
responses are necessary. With a stimulator, there is often a
visible contraction of the pelvic floor. The BCR potentials have
a biphasic appearance (Figure 12). The latency of the
potential is variable: mean = 35.9 msec, with a range of 26 to
44 msec. The BCR potential allows electrophysiological
evaluation of cauda equina and conus medullaris function. That
is, prolongation-or, more commonly, absence-of the BCR potential
suggests dysfunction of the cauda equina and/ or conus
medullaris. Thus, both SSEP and BCR studies can indicate
function of some of the neural pathways traversing the cauda
equina and conus medullaris, two regions frequently involved in
tethered cord syndrome.
Pelvic Floor Electromyography
Another means of evaluating the
child with tethered cord syndrome is EMG of the perineal floor
muscles. This technique usually involves placing a concentric
needle electrode into the external urethra sphincter muscle.
Individual motor unit potentials are examined. 5tandard criteria
for firing rates and other features are used to determine
whether the muscles are normal or denervated. 5ince many pelvic
floor muscles are innervated by different sacral roots, careful
and thorough EMG examination of several muscles may be necessary
to determine the extent of the lesion. Other muscles (in
addition to the external urethra sphincter muscle) that can be
sampled include the external anal sphincter muscles, the
bulbocavernosus muscle, and the ischiocavernosus muscle. This
sort of examination is best performed by a physician, usually a
urologist, with special training in the technique. While EMG can
be performed in a patient of any age, it is usually not done in
infants or very young children unless there is a critically
important diagnostic question regarding the integrity of the S2,
S3, and S4 roots.
Intraoperative Electromyographic Monitoring for Tethered Cord
Syndrome
The lumbosacral anatomy in patients with tethered cord syndrome
is frequently complex. Nerve roots may be embedded in lipoma or
may be visually indistinguishable from adhesions or a thickened
filum terminale. The S1
and lumbar roots are recognizable by palpation of the
contracting muscles after intraoperative electrode stimulation,
but identification of the lower sacral roots requires some
objective means of measuring sphincteric function.
James et al utilized intraoperative external anal sphincter
muscle EMG in 10 patients with spinal dysraphism: four patients
with tethered cord syndrome, three with lipomeningocele, and
three with other miscellaneous diagnoses. These children ranged
in age from 3 weeks to 15 years. Using general anesthesia with
the patient in a prone position, an anal plug or catheter
containing an electrode or needle electrodes was placed in the
anal sphincter muscle for recording purposes. During
dissection, tissues suspected of containing nerve roots close to
the conus were stimulated. If contraction of the anal sphincter
muscle occurred, meticulous care was undertaken to preserve
these structures. No patient deteriorated postoperatively and
two noted improvement.
Pang modified this technique by directly recording the "squeeze
pressure" using a pressure-sensitive balloon inserted into the
anal canal. There is a rationale for using "squeeze pressure":
it had earlier been noted that there is a direct relationship
between anal sphincter muscle integrated EMG and anal canal
pressure measurements with an anal balloon. Pang used a
double-lumen balloon catheter ordinarily used for intraluminal
angioplasty. The balloon does not deform at high pressures so
there is a high degree of sensitivity. The stimulation was done
with a disposable monopolar nerve locator stimulator ring and
three current intensities: 0.5 (usually sufficient for infants
and small children), 1.0, and 2.0 mA. If a sacral root was
stimulated and the external anal sphincter muscle contracted,
the combined stimulus artefact and spike wave on the pressure
tracing was easily recorded. During stimulation, the
cerebrospinal fluid had to be continuously suctioned to prevent
current dispersion. Pressure responses with unilateral S2, S3,
or S4 root stimulation generally generated pressures >40-75 torr,
even in plantar flexion of the foot without pressure change.
Stimulation of the filum terminale and nonneural tissues always
produced stimulus artefact without a pressure wave. Pang
found this technique useful in several circumstances: 1)
identifying sacral roots embedded in intradural lipoma; 2)
identifying the junction between functional conus and
intramedullary lipoma; 3) differentiating an elongated conus
medullaris from a thickened filum terminale; and 4) identifying
thickened adhesions (from previous myelomeningocele repair) and
sacral roots. The more severely impaired the child or the more
complex the disorder, the more the monitoring is needed to
prevent nerve root injury, but also the more difficult it is to
get satisfactory tracings because the sphincter muscles are
paralyzed, and arachnoiditis makes the recording procedures
technically much more difficult. In summary, the S2, S3, and S4
roots, and the conus can be differentiated from the S1 and
lumbar roots, the filum, lipoma, fibrous adhesions, and other nonfunctional fibroneural bands.
Summary
There are several neurophysiological techniques available to the
clinician to aid in the diagnosis and management of the patient
with tethered cord syndrome: SSEP-PTN, SSEPPuN, urodynamics, and
EMG. An understanding of the developmental anatomy, the
functional anatomy of evoked potentials, EMG, and urodynamics
enhance the ability to care for these patients
|
 |
 |
|
Prof. Munir Elias
Our brain is a mystery and to understand it, you
need to be a neurosurgeon, neuroanatomist and neurophysiologist.
neurosurgery.tv

Please visit this site, where daily neurosurgical activities are going
on.

Inomed ISIS IOM System
|
|
|
|
















