|
|
|
 |
|
|
Confusing anatomy is often encountered during operations on complex dysraphic lesions in the lumbosacral canal. It is common to see nerve roots embedded in lipoma or scar tissue, or they may not be easily distinguishable from fibrous adhesion bands. Sometimes nerve roots that are bundled tightly by abnormally thickened arachnoid can look like a thickened filum terminale. Also, the transition between a functional but structurally deformed conus and an intramedullary lipoma is not always visually apparent. Thus, some objective means to identify the sacral nerve roots and the conus is necessary to ensure preservation of these neuronal structures. In addition, in some cases of complex transitional lipomas, the tip of the conus is tautly suspended by low sacral roots that are short, stout, and fibrotic. An assessment of their functional integrity is useful for determining whether dividing them, in order to complete the untethering process, would lead to unacceptable loss of sphincter function. The first sacral and lower lumbar roots are recognized readily by intraoperative nerve stimulation while palpating for contractions of the respective segmental muscle groups through the surgical drapes. Identification of the lower sacral roots and functional quantification of these roots and their corresponding medullary connections, however, require some objective assessment of perineal sensation and sphincter function.
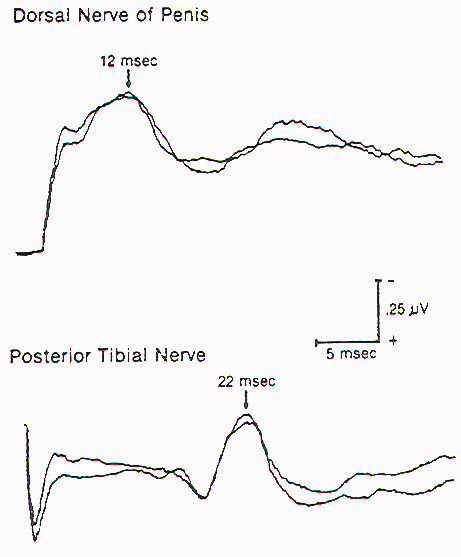
The assessment of evoked responses generated by directly stimulating parts of the sex organs, urethra, and anal canal constitutes the mainstay of sensory monitoring of the lower sacral segments. Monitoring of such responses is most useful when the distal conus or dorsal nerve roots are being rather strenuously handled, as in certain difficult resections of large transitional lipomas or during removal of the median fibrous sleeve of a Type I split cord malformation. The latency and amplitudes of the waveforms are exquisitely sensitive to structural deformation and ischemic changes to the central sensory pathways. Sensory evoked response monitoring is less useful in the identification of sacral sensory roots, because the responses are generated by end organ stimulation. Cortical responses generated by direct dorsal root stimulation give much less predictable waveforms, which are not stable enough for foolproof identification purposes.
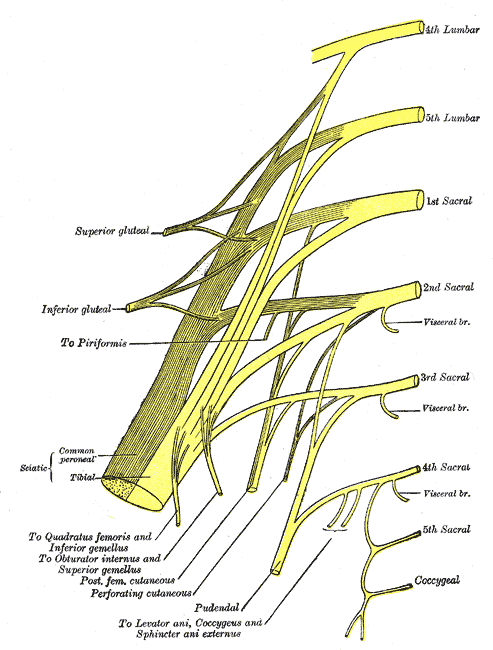
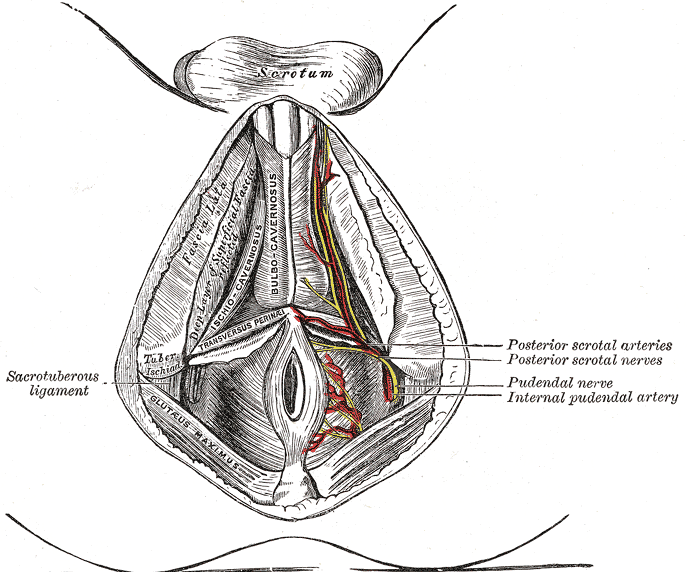
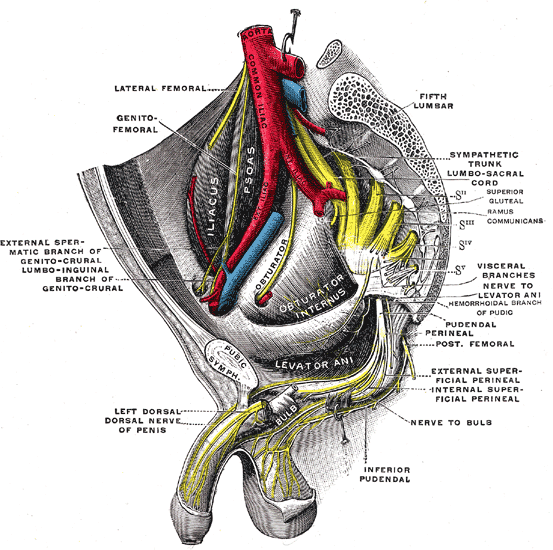
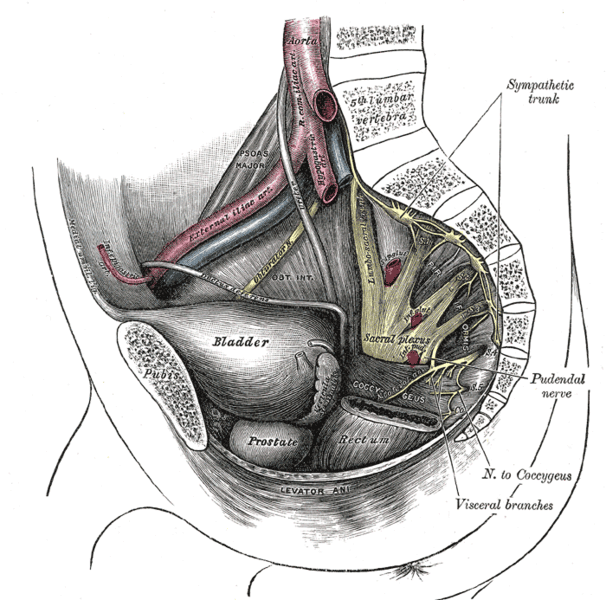
The peripheral nerves that supply the bladder, anal canal, and perineal skin, all potentially available for stimulation, are divided into three main groups. 1.The pudendal nerve is the primary somatic nerve to this region. The pudendal motor neurons innervating the external sphincter and pelvic floor originate from Onuf's nucleus in the anterior horn of the S2 to S4 cord segments. The sensory fibers come from the corresponding dorsal root ganglia. The mixed fibers course via the S2, S3, and S4 roots to exit the spinal canal through the sacral foramina (Figure-1). Somatosensory impulses travel in this nerve from receptors located in the skin of the genitalia and perineum, the pelvic floor, and bulbocavernosus muscles, as well as in the mucosa of the distal urethra and anus. Motor fibers in the pudendal nerve innervate the bulbocavernosus muscle, external urethral sphincter, external anal sphincter, and pelvic floor muscles. The pudendal nerve is the most easily accessible nerve for evoked response testing.
Fig-1: Schematic representation of the pudendal nerve and branching. 3.The pelvic splanchnic nerves supply the sacral parasympathetic innervation to the pelvic organs. The motor neurons in this nerve originate in the S2 to S4 cord segments, slightly more caudal than the pudendal motor neurons. The fibers are distributed to the pelvic organs via the S2 to S4 nerve roots and inferior epigastric plexus. The pelvic nerve carries sensory afferents from the proximal urethra, bladder wall, prostate, seminal vesicles, and rectum. Motor innervation is primarily to the detrusor muscles, the corpus cavernosus, the rectum, and probably the upper smooth-muscle portion of the external urethral sphincter. Evoked responses can be elicited on stimulation of the proximal urethra and bladder, presumably due to activation of the pelvic sensory fibers. 3.The hypogastric nerve plexuses carry autonomic (sympathetic) fibers from the intermediolateral cell column of the T11-L2 spinal cord segments. The preganglionic fibers course via the paravertebral sympathetic chain ganglia, inferior mesenteric plexus, superior hypogastric plexus, and finally the inferior hypogastric plexus. The postganglionic fibers are distributed to the smooth muscles of the bladder neck, the smooth-muscled internal urethral sphincter, the parasympathetic intramural ganglia of the detrusor muscles and probably the intrinsic portion of the external urethral sphincter. The postganglionic fibers also share connections with plexuses around the rectum and anal canal, seminal vesicles, ductus deferens, prostate, and corpus cavernosus in the male, and vagina in the female. It is uncertain how much the afferent component of the hypogastric nerves contributes to the evoked response in humans.
Pudendal sensory evoked responses are useful in monitoring intraoperative injury to the conus and lower sacral sensory nerve roots, but they are neither qualitatively nor quantitatively suitable for the identification of the lower sacral roots (especially the motor roots) or conus from non-neural elements. Intraoperative identification requires some way of measuring the oneto-one stimulus-to-response coupling of end organ function when the nerve root in question is being stimulated. For the lower sacral roots, this means the assessment of sphincter function.
External anal sphincter electromyography (EMG) has long been found useful as a qualitative tool for studying anorectal closure function and disorders. The EMG electrodes are either embedded in an anal plug or anal balloon, which is placed into the anal canal, or are in the form of needles inserted directly into the external anal sphincter transmucosally. The needle electrodes are more reliable because they are not subject to dislodgement or to having mechanical artifacts during contraction of the sphincter itself; however, accurate and secure placement of the needles requires some expertise and is initially best done by the neuro-urologist. The grounding plate is pasted on the patient's thigh, and EMG recordings are made using a standard bladder diagnostic unit. The sensitivity of the recording stylus is adjusted so that minimal deflection occurs at rest. With stimulation of the lower sacral motor roots, the stylus gives a discrete one-to-one spike-deflection, much different than the baseline.
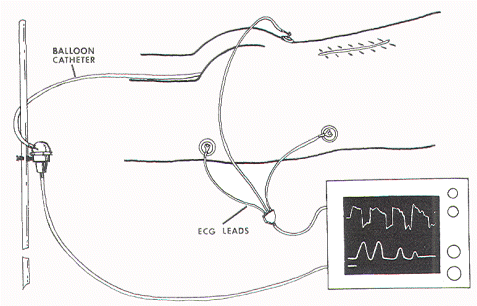
The external anal sphincter EMG requires bulky and expensive equipment, as well as the availability of someone expert in the accurate placement of the needle electrodes. An alternate method of monitoring anal sphincter function is the direct measurement of the "squeeze pressure" induced by sacral root stimulation using a pressure-sensitive balloon inserted in the anal canal. This technique is simple and noninvasive, requires no special expertise, utilizes inexpensive, portable equipment, and produces easily interpretable pressure waves which are semi-quantitative and virtually unaffected by other electronic components in the operating room that are known to cause annoying baseline noise in an EMG recording.
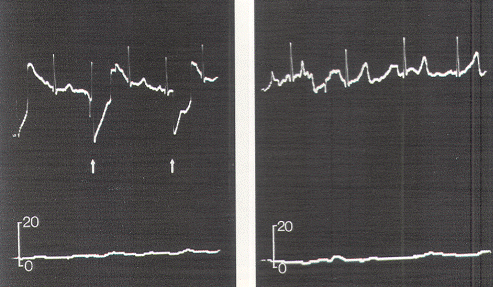
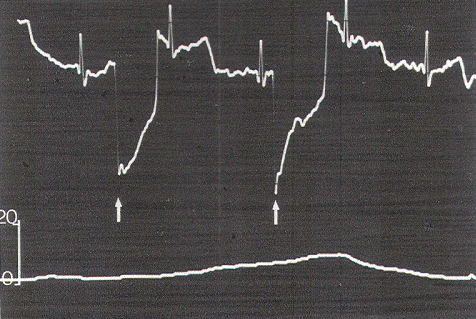
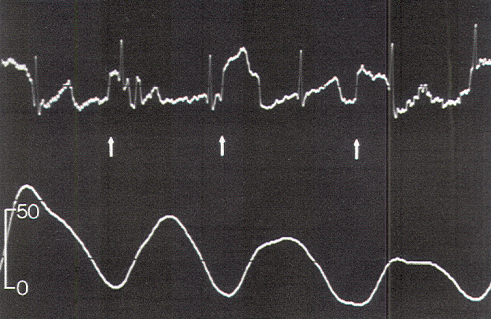
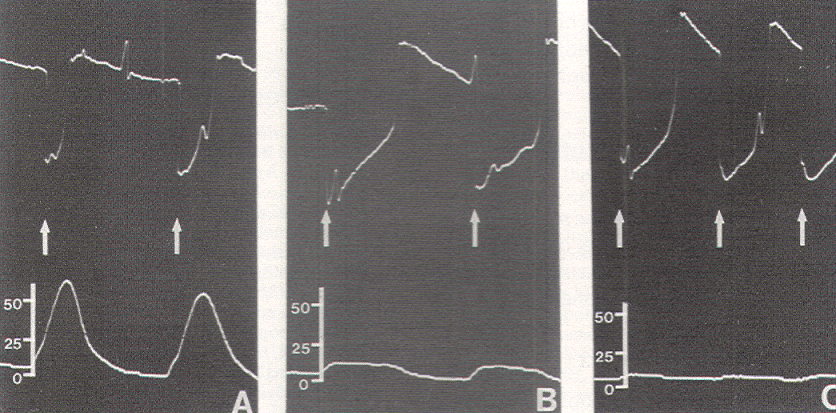
The relationship between EMG and contractile strength in a longitudinal muscle was first defined by Lippold, who found a linear relationship between the integrated action potentials on the EMG and the tension generated by voluntary isometric contractions of the human gastrocnemius. This linearity was explained by the fact that an increase in contractile strength of a muscle is brought about either by a spatially random increase in the number of contracting motor units or by random increments of discharge frequencies of the active units; in both situations, the integrated electrical output of the muscle would increase proportionately. The same linear relationship was also demonstrated in the external anal sphincter by Schweiger, who made simultaneous recordings of sphincter EMG and anal canal pressures with an anal balloon. These data support the validity of using squeeze pressure, instead of sphincter EMG, to monitor the functional status of the lower sacral motor neurons. In order for the anal pressure monitor to be operational, some sphincter function must be present. Theoretically, a severely damaged motor nerve with only enough viable axons to generate a barely visible EMG would produce no measurable squeeze pressure; in such a situation, the EMG might be more sensitive. However, such a nerve would not provide useful sphincter function for the patient, and its preservation is of doubtful value. In the author's experience, any external anal sphincter that could generate enough voluntary or reflex (as in the bulbocavernosus or anal wink reflex in the infant) contractions to be appreciable by preoperative digital examination should produce recognizable pressure spikes on the anal pressure monitor. This anal balloon monitor is therefore sufficiently sensitive for the practical purpose of sacral root and conus identification.
The external anal sphincter consists of a bulky deep part, a fusiform superficial part, and a subcutaneous part decussating behind and in front of the anus. It encloses the lower part of the levator ani, the anorectal junction, and the anal canal in the shape of a funnel. The internal anal sphincter arises from the muscular coats of the rectum and insinuates itself between the rectal mucosa and the upper portion of the funnel. The external anal sphincter is innervated by the pudendal nerve. This arises from the anterior division of S2 and S3 and both divisions of S4, enters the pudendal (Alcock's) canal through the lesser sciatic foramen, and divides into two main branches just proximal to the urogenital diaphragm. The proximal branch, the inferior hemorrhoidal nerve, supplies the striated muscles of the external anal sphincter; the distal branch, the perineal nerve, supplies the external urethral sphincter. The internal anal sphincter, composed of smooth muscles, is innervated by the hypogastric nerve, derived from the intermediolateral (sympathetic) columns of L1 and L2. Stimulation of the S2, S3, and S4 roots, therefore, activates only the external and not the internal anal sphincter. Furthermore, unless there is localized disease or trauma to the pudendal branches at the urogenital diaphragm, activity of the external anal sphincter reflects function of the external urethral sphincter. The anal pressure balloon described here is an elongated ellipsoid selected specifically to pick up activities from all three parts of the external sphincter funnel. Its elongated span also minimizes the possibility of accidental dislodgement by contractions of the pelvic musculature induced intraoperatively. Although the elongated balloon will also pick up contractions of the internal anal sphincter, the latter is never activated by the nerve stimulator or by manipulation of the lower sacral spinal cord or nerve roots because its nerve supply is from L1 and L2. However, being made up of smooth muscles, the internal sphincter does have spontaneous rhythmic contractions that will be registered by the balloon, and these must be distinguished from the stimuli-generated pressure spikes from the external anal sphincter.
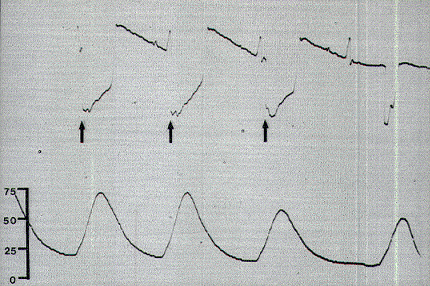
Occasionally, a small pressure wave of less than 7 torr follows S1 stimulation (which does not innervate the sphincters) because of compression on the protruding proximal portion of the balloon by the medial inferior fibers of the gluteus maximus. Although such "ripple waves" are easily differentiated from the tall spike waves of healthy lower sacral roots, they could be mistakenly construed as the subdued responses seen with partially damaged S2, S3, and S4 roots. This confusion is eliminated if care is taken to secure the posterior end of the balloon just above the mucocutaneous junction. Stimulation of the filum terminale and nonneural tissues always produces a stimulus artifact but not a pressure wave. Thus, the S2, S3, and S4 roots and the conus can be distinguished from the S1 and lumbar roots, the filum, lipoma, fibrous adhesions, and other nonfunctional fibroneural bands, such as an occult myelomeningocele. The ECG artifact and pressure wave relationships are summarized in Table 1.
Needle recording electrodes can be percutaneously inserted into the "extrinsic" portion of the external urethral sphincter to monitor activity of this sphincter. This technique is routinely used by neurourologists to correlate simultaneous measurements of bladder pressure, urethral pressure, and external urethral sphincter activities. Pelvic floor EMG can thus be used for intraoperative sacral root identification in the same manner as external anal sphincter EMG.
Two reflexes with centers in the sacral cord can be utilized to assess the integrity of both the sensory and motor roots as well as their interconnecting intramedullary components.
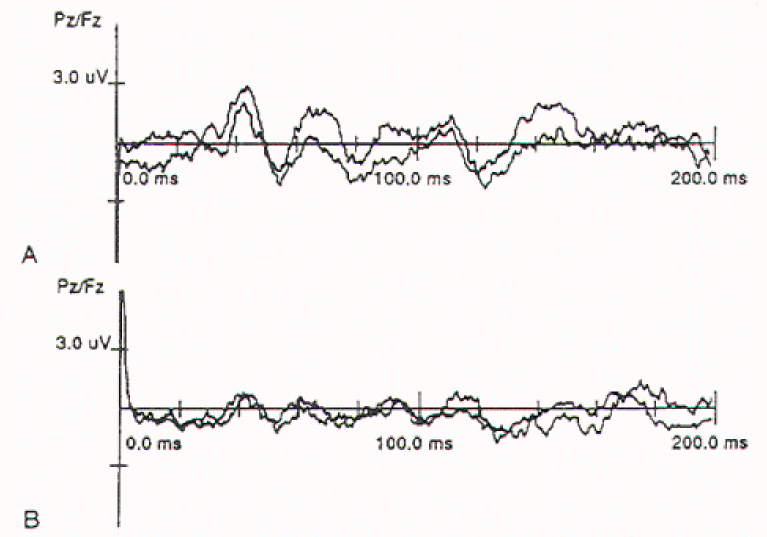
The reflex response of the bulbocavernosus muscle to stimulation of penile nerves can be studied using square wave electrical stimuli applied through ring electrodes on the penis, and recorded either by needle electrodes in the muscle or by surface electrodes fixed to the midline of the perineum, between the base of the penis and the anus. The averaged response from 50 to 100 stimuli is usually biphasic with an initial negative peak. The latency for most healthy adults is 24 to 42 msec but varies with age and maturation in young children. The waveform is also distorted significantly in most cases of myelodysplasia and tends to become "unstable" with very minor manipulations of the conus. The use of this monitoring modality is therefore limited and is feasible only in patients with virtually normal sphincter function preoperatively.
The urethral to anal sphincter reflex can be measured using stimulating electrodes similar to those used in eliciting urethral cortical evoked responses and recording electrodes used in recording external anal sphincter EMG. The latency is considerably longer (50 to 70 msec) than the bulbocavernosus reflex, although their morphologies are similar. The long latency in the urethral-anal sphincter reflex is due partly to the slower conducting velocity of autonomic afferent fibers and partly to a more complex central polysynaptic reflex organization.
|
|
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Copyright [20016] [CNS Clinic-Jordan]. All rights reserved